Cauta Produse si Servicii
de la peste 100.000 de firme sau PFA-uri din Romania

de la peste 100.000 de firme sau PFA-uri din Romania

Cumpara produse sau servicii de la prestatori sau producatori directi, recunoscuti in Romania.
Promovam peste 500.000 de produse sau servicii de la peste 100.000 de firme si PFA-uri.
Inregistreaza-te pe Bizoo.ro si lasa clientii multumiti sa iti promoveze gratuit afacerea. Din Gura in Gura!

Bizoo a creat site-uri pentru peste 100.000 firme din Romania.
50% din firme nu au inca un site web. Firma ta are unul?
Adauga adresa site-ului tau pe carti de vizita si materiale promotionale. Simplu!
Produsele tale sunt promovate imediat in fata a peste 100.000 de firme din Romania.
Bizoo poate prelua produsele din site-ul tau cu ajutorul unui DataFeed.

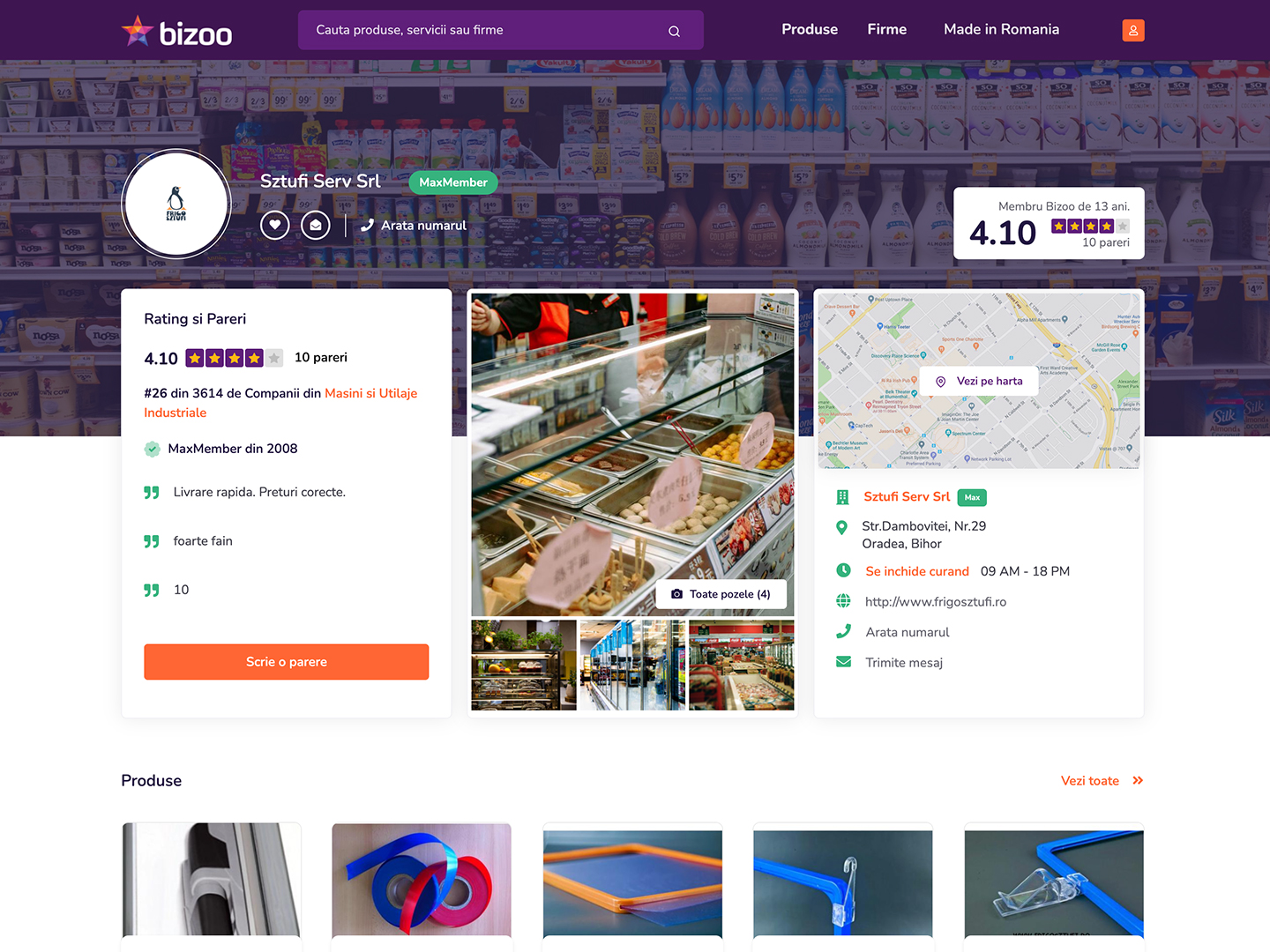
De acum, poti afisa ratingul si parerile clientilor tai oriunde ai ocazia.
Vanzarile firmei tale vor creste ridicol de rapid. Din Gura in Gura.

 5/5
5/5
"Felicitari echipei care s-a adaptat permanent situatiei economice, a modernizat utilajele si le-a eficentizat!
In decursul timpului, am cercetat piata, atat firmele concurente cat si clientii care deja au cumparat si utilizat utilaje de procesare a hartiei si carton.
Ca si concluzie, recomand cu incredere tuturor celor interesati produselor concepute si realizate de echipa Lemar Impex SRL, recomandate de seriozitatea si profesionalismul lor!"

 5/5
5/5
"Cei mai buni , serioși și cu cea mai mare calitate , sunt fără dar și poate cei de la Adriano! Este mai mare dragul sa mi comand haine de la ei! Iar de personal nici nu mai zic! Doamna care Răspunde la telefon este pur și simplu o dulce! Mulțumim Adriano! Pe lângă faptul ca sunteți cei mai tari, sunteți și unici! Oamenii faini! ❤️"

Un MaxMember are cu 950% mai multi potentiali clienti decat un Membru Gratuit. Garantat!
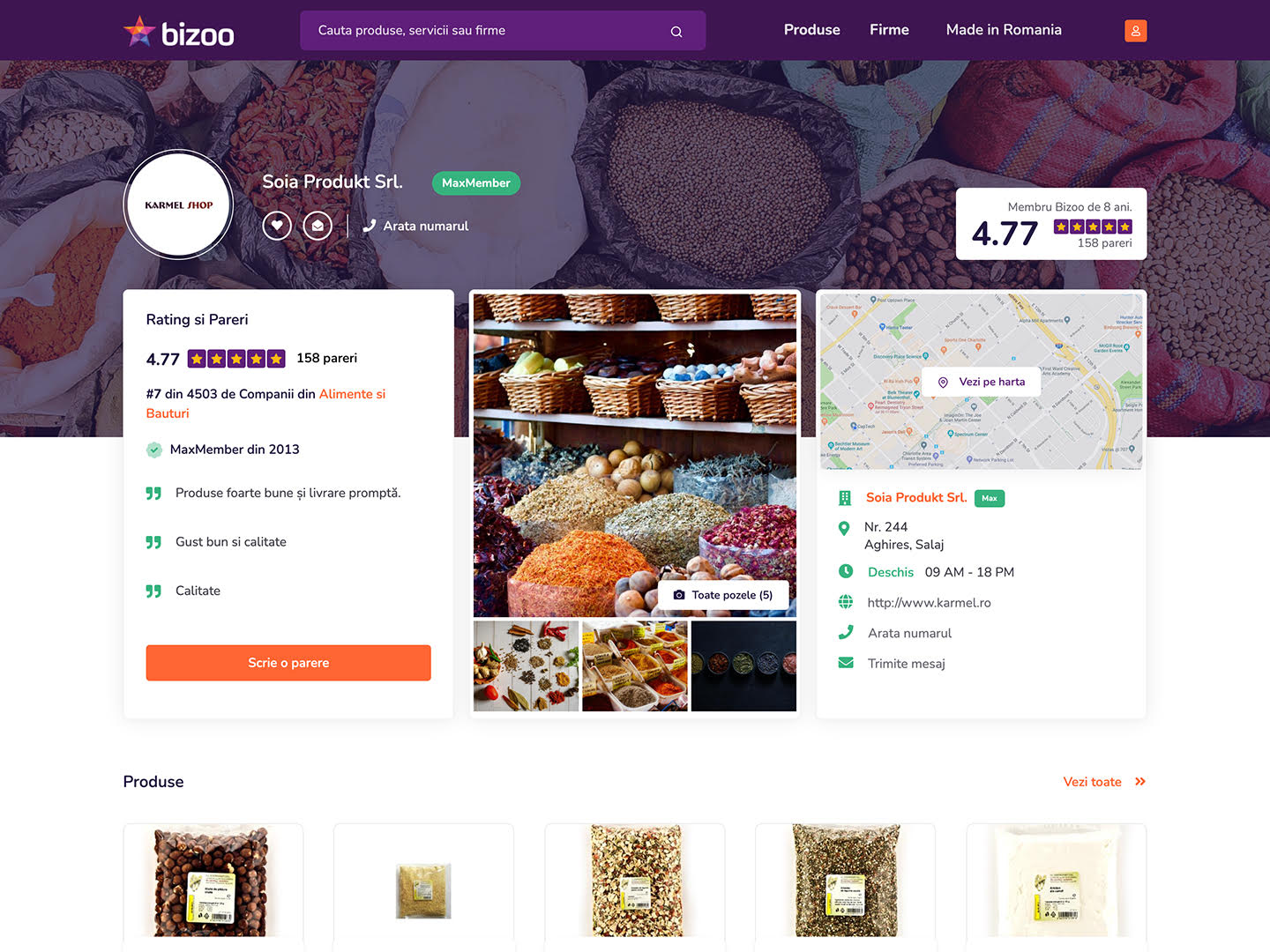
Produsele unui MaxMember sunt primele, mereu in fata Membrilor Gratuiti
Un MaxMember are de pana la 1000x mai multe produse sau servicii la vanzare
Cu Bizoo ai un site modern, simplu de personalizat, adaptat pentru mobil
Clientii nostri sunt fanii nostri si in timp, au devenit prietenii nostri.